Challenge
Medical research projects involve the use of highly sensitive data that must be shared across multiple departments. Ensuring data privacy, preventing unauthorized re-linking, and complying with regulatory guidelines are paramount, all while maintaining the data's utility for machine learning and analytical purposes.
Solution
Anonos
Data Embassy offers a comprehensive suite of Privacy-Enhancing Technologies (PETs) designed to safeguard sensitive healthcare data while preserving its utility. These techniques can be meticulously tailored to suit each stage of data processing and the specific needs of the project:
- Statutory Pseudonymization: This technique protects both direct and indirect identifiers in the data by using dynamic tokens to prevent unauthorized re-linking. This approach maintains the data's analytical utility and allows for the reconnection of data to the original records, but only by authorized personnel under controlled conditions.
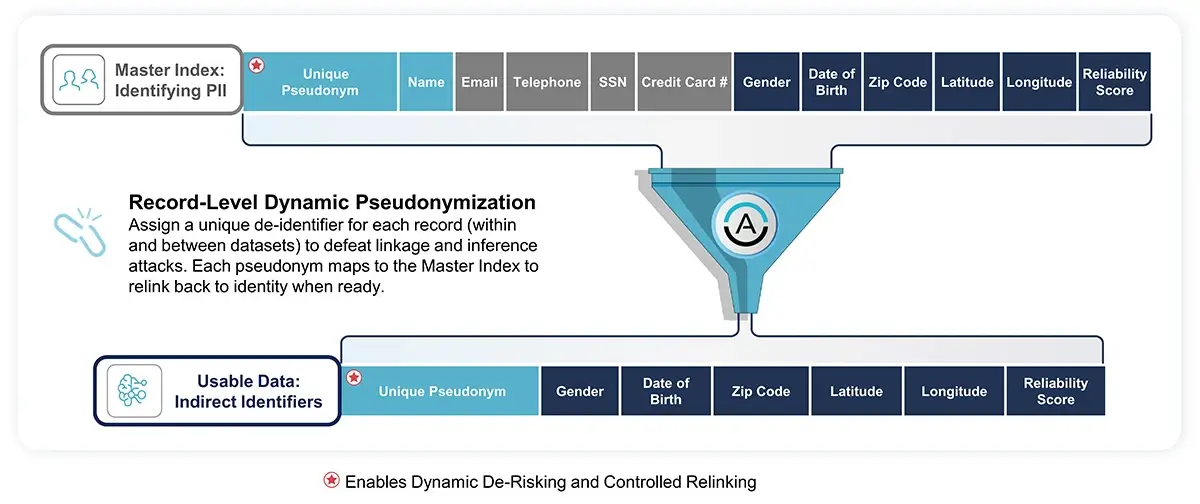
-
Synthetic data: A significant challenge in many research projects is the scarcity of sufficient data to formulate balanced and unbiased hypotheses. Synthetic data technology addresses this by producing artificial data records to complement and balance original pseudonymized datasets. This approach not only augments the dataset with anonymized synthetic healthcare data but also empowers scientists to conduct comprehensive exploratory analyses without compromising privacy.
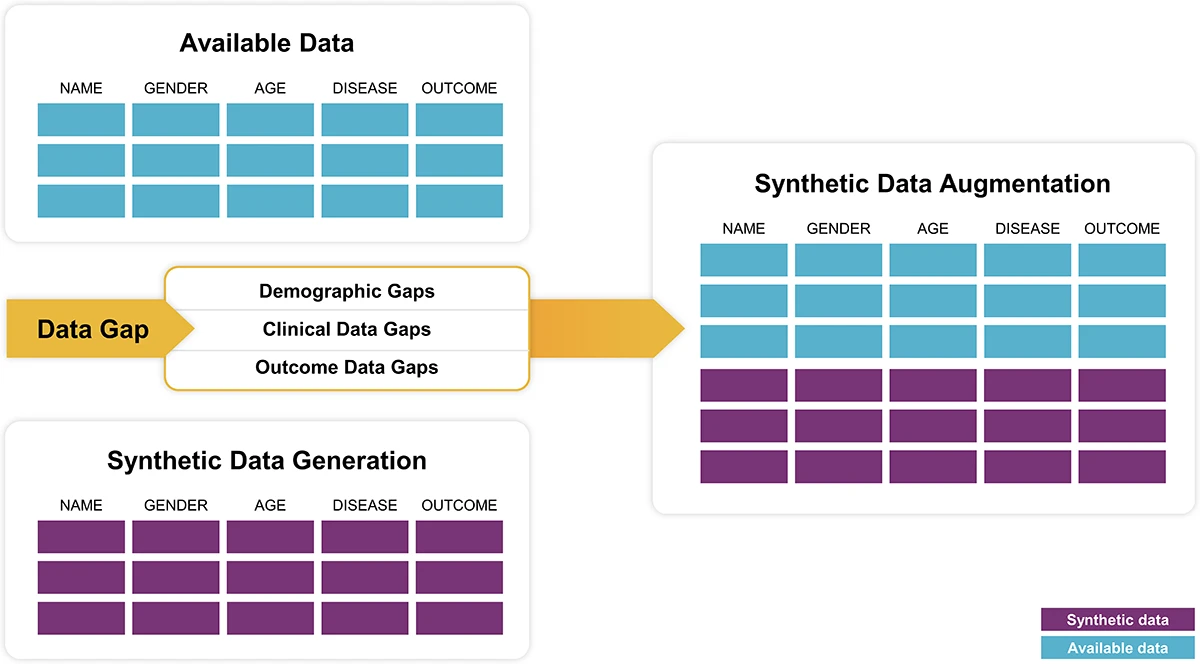
Results
Regulatory Compliance
Successfully meet compliance with global and local data protection regulations, mitigating the risk of penalties.
Operational Efficiency
Reduce healthcare data preparation and compliance validation times, streamlining the overall research process.
Accelerate Time-to-Insight
Streamlined data security and privacy procedures enable a faster time-to-insight for researchers, leading to quicker hypothesis validation.




